What’s Driving Life Science Literature Reviews?
Systematic literature reviews (SLRs) are the backbone of evidence-based decision-making in life sciences. But not every SLR is submission-related. So, what purpose do these rigorous reviews serve beyond journal publication or HTA submission?
CapeStart’s 2024 Literature Review Research Report offers valuable insights into this question, with responses from 79 life sciences professionals actively involved in literature reviews. The data reveals a dynamic landscape in which Health Economic and Outcomes Research (HEOR), Market Access (MA), and Real-World Evidence (RWE) professionals use SLRs to serve multiple critical functions, from regulatory to strategic.
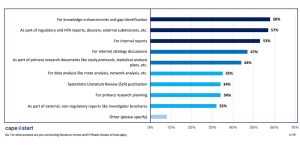
- Identifying Knowledge Gaps—Topping the list, 58% of respondents conduct literature reviews to identify and address gaps in existing scientific knowledge. For these respondents—in HEOR, MA, and RWE—understanding what’s missing is just as important as what’s already known, especially when developing models or identifying endpoints for economic evaluations.
- Supporting Regulatory and HTA Submissions—A similar proportion (57%) leverages literature reviews for regulatory and health technology assessment (HTA) submissions. Well-structured SLRs are essential to demonstrate the value, effectiveness, and cost-efficiency of treatments to payers and regulatory bodies.
- Creating Internal Reports—More than half (53%) of respondents rely on SLRs to inform internal decision-making. Whether assessing a therapy’s market landscape or guiding pipeline development, internal reports backed by comprehensive literature reviews help stakeholders align their strategies with evidence.
- Informing Strategy and Primary Research—Literature reviews are also vital tools for internal strategy discussions (47%) and primary research planning (34%). Life science professionals often use SLR findings to refine research questions, define study populations, or support economic model inputs with the most relevant, up-to-date data.
- Feeding Data Analysis & Publications—A significant number of professionals use literature reviews for data analyses—including meta-analyses and network meta-analyses (35%)—and for generating SLR publications (34%). To demonstrate evidence, literature reviews are required for publication in most life science journals.
- Supporting Clinical Documents—SLRs also play a supportive role in drafting clinical materials such as study protocols and statistical analysis plans (37%). In HEOR, this translates to ensuring that clinical assumptions align with current evidence, improving the robustness of economic modeling and outcomes research.
Respondents share that they are using SLRs as strategic instruments—guiding research, satisfying regulatory expectations, enriching internal planning, and reinforcing the evidence base for economic decision-making. As healthcare continues to evolve toward value-based care and data-driven insights, the demand for agile, accurate, and purpose-driven literature reviews will only grow. HEOR, MA, and RWE professionals sit at the intersection of clinical evidence and economic justification—making their use of SLRs both diverse and essential.
To dive deeper into the full findings, check out CapeStart’s 2024 Literature Review Research Report.

